Vaccination available at our office. Make an Online Appointment for Flu Vaccination to come in at your convenience, or call (877) 696-9263. Supplies are limited and appointments are required.
Much of the illness and death caused by the flu can be prevented by a yearly flu shot. People in high-risk groups and people who are in close contact with those at high risk should get a flu shot every year. Influenza, or the flu, is generally not dangerous, but complications from the infection can be. The flu vaccine (a flu shot), creates an immunity to the flu and protects you against the flu. The flu shot is made from inactivated viruses (the viruses are killed), and experts consider the flu shots safe. Make an appointment for Flu Vaccination and Consultation today. We use Fluzone® from Aventis®.
Who Should Get a Flu Shot?
Some people who get the flu become seriously ill and need to be hospitalized. Some people even die from illnesses related to the flu. That’s why it’s important for you and your doctor to determine whether your family needs the flu vaccine.
- Adults and children (> 6 months) who want to avoid the flu
- Adults and children (> 6 months) with chronic heart or lung conditions & asthma
- Persons 50 years or older

- Adults and children (> 6 months) who need regular medical care or had to be in a hospital because of metabolic diseases (like diabetes), chronic kidney disease, or weakened immune system
- Residents of nursing homes and other long-term care facilities
- Children and teenagers (6 months to 18 years) who are on long-term aspirin therapy
- Women who will be more than 3 months pregnant during the flu season
- Persons who provide important community services (such as police, fire department personnel, emergency medical services, doctors, nurses and hospital workers).
The flu vaccine reduces the average person’s chances of catching the flu by up to 80% during the season. For healthy young adults, the flu shot reduces the risk of getting the flu by up to 90%. Even if an immunized person gets the flu, symptoms usually will be fewer and milder.
Is the Flu Vaccine Safe During Pregnancy & While Breastfeeding?
Pregnancy can increase the risk for complications from the flu, and pregnant women are more likely to be hospitalized from complications of the flu than non-pregnant women of the same age. Women who will be beyond the first 3 months of pregnancy during the flu season should get a flu shot. Pregnant women who have medical problems that increase their risk for complications from the flu should get a flu shot before the flu season, no matter their stage of pregnancy. Also, it is safe to get a flu shot if you are breastfeeding. A flu shot cannot cause flu in either you or your baby.
When Should You Get a Flu Shot?
We are now offering the flu vaccine, and recommend getting vaccinated between September and mid-November (but you may be given the vaccine at other times of the year). Flu season is from November to April, with most cases occurring between late December and early March. The best defense is get vaccinated as soon as the vaccine becomes available.

The Vaccine Does Not Cause the Flu.
A common myth about the flu shot is that it can actually cause the flu. But the flu vaccine in the United States is made from killed influenza viruses, which means that it’s impossible to catch the flu by getting this vaccine. Each year, the vaccine is updated to include the most current strains of the virus. After getting a flu shot, a person’s body will create antibodies to fight the virus if exposed to it. Antibodies against flu develop and provide protection within 1 or 2 weeks after vaccination.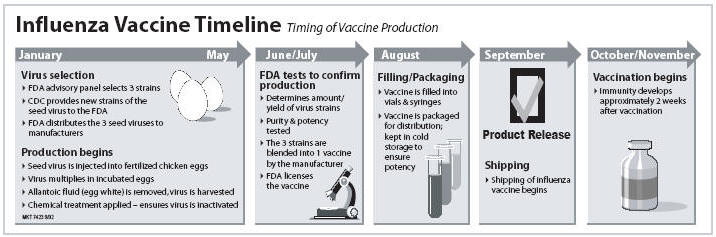
Inactivated (killed-virus) influenza vaccine containing antigens identical or similar to currently circulating influenza A and B viruses has been shown in controlled trials to be 70-80% effective in preventing influenza illness or reducing severity of influenza illness in healthy children, adolescents, and adults under age 65.1-5 The vaccine has also been reported to reduce clinical symptoms in health care workers,6 which may translate into a reduction in transmission to high-risk patients.
Influenza vaccine should be administered annually to all persons ages 65 and older and to persons 6 months of age or older who are residents of chronic care facilities or suffer from chronic cardiopulmonary disorders, metabolic diseases (including diabetes mellitus), hemoglobinopathies, immunosuppression, or renal dysfunction (“B” recommendation). Influenza vaccine is also recommended for health care providers for high-risk patients (“B” recommendation).

In persons at high risk for influenza A (e.g., during institutional outbreaks), amantadine or rimantadine prophylaxis (200 mg/day orally) may be started at the time of vaccination and continued for 2 weeks (“B” recommendation). A lower dose (less than or equal to 100 mg/day) of amantadine is recommended for persons with reduced creatinine clearance and those 65 years of age and older. A reduced dosage (100 mg/day) of rimantadine is indicated for those with reduced renal or hepatic function and for elderly nursing home residents and may also be necessary in healthy persons 65 years and older who experience side effects. Amantadine and rimantadine are most useful as short-term prophylaxis for high-risk persons who have not yet received the vaccine or are vaccinated after influenza A activity in the community has already begun; when the vaccine may be ineffective due to major antigenic changes in the virus; for unimmunized persons who provide care for high-risk persons; to supplement protection provided by vaccine in persons who are expected to have a poor antibody response; and for high-risk persons in whom the vaccine is contraindicated (i.e., those with anaphylactic hypersensitivity to egg protein). If vaccine is contraindicated, amantadine or rimantadine should be started at the beginning of the influenza season and continued daily for the duration of influenza activity in the community.
Make an Appointment for Flu Vaccination
